Journal of Clinical Nuclear Medicine Publishes Results of Tiziana’s Nasal Foralumab in Study Treating Moderate Alzheimer's Disease
This case represents the first use of 18F-PBR06-PET in a patient with moderate AD dementia and the first published report of an AD patient treated with nasal-foralumab. The study, led by Dr.
Figure 1.
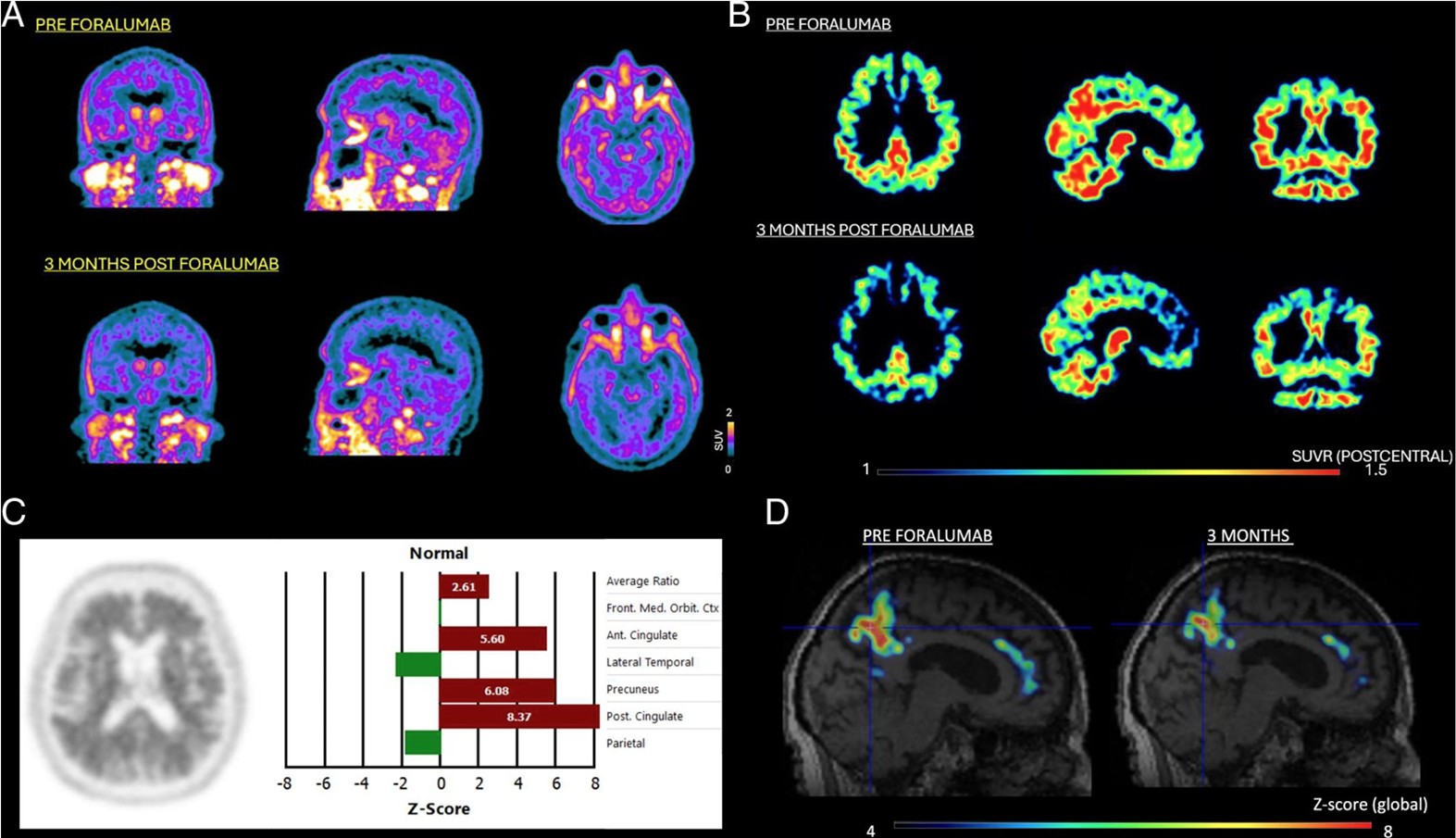
Image Source: Singhal T et al. Dampening of Microglial Activation with
A, 18F-PBR06-PET shows significant decreased standardized uptake value (SUV) after 3 months of treatment (bottom row) with nasal-foralumab as compared to baseline (top row).
B, SUV-ratio (SUVR) images demonstrate a decrease in PET signal following treatment, particularly in bilateral precuneus, posterior cingulate, bilateral parietal cortices and cerebellum (bottom row) as compared with pre-treatment baseline (top row).
C, A baseline amyloid-positive 18F-Florbetapir-PET scan in the patient
D, z-score maps of 18F-PBR06-PET focusing on the precuneus, posterior cingulate, and anterior cingulate regions show abnormally increased microglial activation in these regions (left) before treatment, which decreased following treatment with nasal-foralumab (right).
“The marked decrease in microglial PET signal following nasal-foralumab treatment provides preliminary evidence that intranasal foralumab can modulate microglial activity in Alzheimer's disease. This supports the potential efficacy of nasal foralumab in targeting microglial activation in AD,” said
“These findings from
Foralumab is the only fully human anti-CD3 monoclonal antibody in clinical development for intranasal delivery. By modulating T cell function, it aims to reduce inflammation in the central nervous system. Previous studies have shown its potential in multiple sclerosis and other neuroinflammatory conditions.
The full study is available online in Clinical Nuclear Medicine at: https://journals.lww.com/nuclearmed/Fulltext/9900/dampening_of_microglial_activation_with_nasal.1721.aspx.
About Foralumab
Foralumab, a fully human anti-CD3 monoclonal antibody, is a biological drug candidate that has been shown to stimulate T regulatory cells when dosed intranasally. At present, 10 patients with Non-Active Secondary Progressive Multiple Sclerosis (na-SPMS) have been dosed in an open-label intermediate sized Expanded Access (EA) Program (NCT06802328) with either an improvement or stability of disease seen within 6 months in all patients. In addition, intranasal foralumab is currently being studied in a Phase 2a, randomized, double-blind, placebo-controlled, multicenter, dose-ranging trial in patients with non-active secondary progressive multiple sclerosis (NCT06292923).
Foralumab is the only fully human anti-CD3 monoclonal antibody (mAb) currently in clinical development. The non-active SPMS intranasal foralumab Phase 2 trial (NCT06292923) began screening patients in November of 2023. Immunomodulation by intranasal foralumab represents a novel avenue for the treatment of neuroinflammatory and neurodegenerative human diseases.[1],[2]
About
For more information about
Forward-Looking Statements
Certain statements made in this announcement are forward-looking statements. These forward-looking statements are not historical facts but rather are based on the Company's current expectations, estimates, and projections about its industry, its beliefs, and assumptions. Words such as 'anticipates,' 'expects,' 'intends,' 'plans,' 'believes,' 'seeks,' 'estimates,' and similar expressions are intended to identify forward-looking statements. These statements are not guarantees of future performance and are subject to known and unknown risks, uncertainties, and other factors, some of which are beyond the Company's control, are difficult to predict, and could cause actual results to differ materially from those expressed or forecasted in the forward-looking statements. The Company cautions security holders and prospective security holders not to place undue reliance on these forward-looking statements, which reflect the view of the Company only as of the date of this announcement. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties related to market conditions and other factors described more fully in the section entitled ‘Risk Factors’ in Tiziana’s Annual Report on Form 20-F for the year ended
For further inquiries:
+44 (0) 207 495 2379
email: info@tizianalifesciences.com
[1] https://www.pnas.org/doi/10.1073/pnas.2220272120
[2] https://www.pnas.org/doi/10.1073/pnas.2309221120
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/cd486638-7caa-40f2-8c97-1c9c48a3ea9e

Figure 1
A, 18F-PBR06-PET shows significant decreased standardized uptake value (SUV) after 3 months of treatment (bottom row) with nasal-foralumab as compared to baseline (top row). B, SUV-ratio (SUVR) images demonstrate a decrease in PET signal following treatment, particularly in bilateral precuneus, posterior cingulate, bilateral parietal cortices and cerebellum (bottom row) as compared with pre-treatment baseline (top row). C, A baseline amyloid-positive 18F-Florbetapir-PET scan in the patient D, z-score maps of 18F-PBR06-PET focusing on the precuneus, posterior cingulate, and anterior cingulate regions show abnormally increased microglial activation in these regions (left) before treatment, which decreased following treatment with nasal-foralumab (right).
Source: Tiziana Life Sciences Ltd.